Activation Energy Can Best Be Described as the
An activated complex can best be described as an a. Activation energy energy of activated complex energy of reactants.

Pin On Biochemistry Blood Cells
Energy of the activated complex.
. Activation energy can be. The minimum amount of additional energy needed by a reacting molecule to get transformed into the product is termed activation energy. Activation energy can be described as the a.
In a reaction the spark plug of an engine can best be classified as A a catalyst B a supplier of activation energy C the rate determining step D the activated complex E the volatile reactant 1422. Reactions will benefit by having the activation energy decreased - it will make the reaction proceed in the forward direction more easily. Answer 1 of 3.
There is a precise compatibility between an enzymes active site and the substrate molecule. 1 10 kJ 3 30 kJ 2 20 kJ 4 50 kJ 21. Consider the two-step reaction below.
1 hydrogen iodide 3 ethyne 2 carbon dioxide 4 ethene 22. Activation energy can be described as the minimal energy which is required to start a reaction. 3White phosphorus has a lower activation energy than red phosphorus.
Can The Activation Energy Be Negative Yes the activation energy be negative. The Activation Energy can best be described as. Group of answer choices a The amount of energy lost in an exothermic reaction.
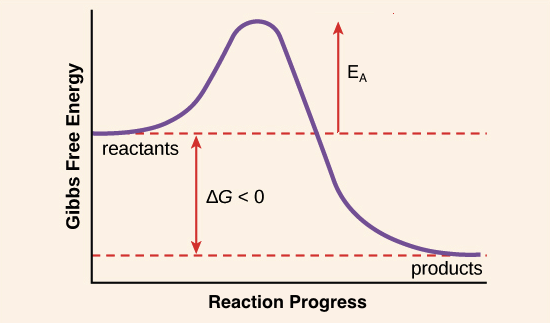
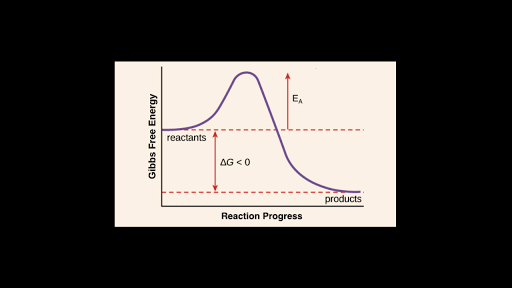
Activation energy can be described as _____. Energy difference between the reactants and the products. The activation energy for the overall reaction is often much greater than the activation energy for the rate limiting step.
Activation energy is the hump that needs to be overcome for a reaction to proceed. Energy difference between the reactants and the activated complex. White phosphorus has a lower activation energy than red phosphorus.
The lower the activation energy would be the more the energy will be available for the reaction to proceed. 4How can the chemical potential energy in an endothermic reaction best be described1 point Reactants have higher chemical potential energy than products. This can be understood by turning once again to the reaction between ClNO 2 and NO.
4Products have higher chemical potential energy than reactants. 2energy stored in chemical bonds. A substance that increases the rate of a reaction without appearing in the equation for the overall reaction is aan A.
A numerical description of the amount of energy needed by colliding reactant molecules in order to form. Reaction where temperature increases and reaction rates decrease can witness negetive activation energy. Activation energy should be added.
The energy threshold that must be reached before a reaction can proceed and products may be formed. The activation energy in the Arrhenius equation can best be described as asked Jun 26 2017 in Chemistry by maju88 a. A reaction can be occurred by reducing the activation energy of the reaction or increasing the activation energy of the reactants.
The reaction of hydrogen and oxygen to form water is best described as 1 exothermic because. White phosphorus has a higher activation energy than red phosphorus. Only a small fraction of the collisions between reactant molecules convert the reactants into the products of the reaction.
What best explains the observation that enzymes are selective in the reactions they catalyze. Unstable arrangement of colliding particles that can rearrange to form products. High-energy complex that is stable enough to isolate and characterize.
None of the answer choices are correct. Usually the activation energy is lowered by the. The total activation energy for the reaction left to right is the difference between Transition State 2.
The activation energy is found by subtracting the energy of the reactants from the energy of the activated complex. Energy of activation The energy of activation is best described as Multiple Choice the speed at which a reaction proceeds to form products. B The amount of energy gained in an endothermic reaction c All of the above d The amount of energy required to start a reaction.
High-energy molecule with strong bonds and a stable structure. Energy needed to activate the reactants and trigger a chemical reaction. The Activation Energy of Chemical Reactions.
Again we can read the energy of the reactants and activated complex off the graph. The distance marked Z represents A The activation energy for Ag Bg Cg Dg B The heat of reaction energy for Ag Bg Cg Dg C The activation energy for Cg. Stable molecule with a long lifetime.
Activation energy is the minimum amount of energy needed to start a chemical reaction. 1energy input needed to break bonds of reactants. A protein that speeds up a chemical reaction.
According to Reference Table I which compound released the greatest amount of energy per mole when it is formed from its elements. Activation Energy kJ ΔH kJ A. Activation energy for the reverse reaction.
Activation Energy Article Khan Academy

Activation Energy Definition Formula Si Units Examples Calculation

Utopian Sustainable Architecture Big Architects Diagram Design Diagram Architecture
Activation Energy Article Khan Academy

Activation Energy Definition Formula Si Units Examples Calculation

Pin On Secrets Of The Universe
Activation Energy And Temperature Dependence Boundless Chemistry

No comments for "Activation Energy Can Best Be Described as the"
Post a Comment